|
Oknina, L. B., Wild-Wall,
N.,
Oades, R. D., Juran, S. A., Röpcke, B., Pfueller, U., Weisbrod,
M., Chan, E., & Chen, E. Y. H.,
(2005). Frontal and temporal sources of mismatch negativity in healthy
controls, patients at onset of schizophrenia in adolescence and others
at 15 years after onset . Schizophrenia
Research, 76,
25-41.
request a copy
In accord with our understanding of journal policy,
we present the
pre-publication text (view).
The final version (cited above: doi:10.1016/j.schres.2004.10.003)
is available at http://www.sciencedirect.com/science?_ob=ArticleURL&_udi=B6TC2-4DS8DBP-
1&_user=10&_coverDate=07%2F01%2F2005&_rdoc=3&_fmt=high&_orig=browse&_srch=doc-
info(%23toc%235158%232005%23999239998%23597342%23FLA%23display%23Volume)&_cdi=5158&_sor
t=d&_docanchor=&_ct=16&_acct=C000050221&_version=1&_urlVersion=0&_userid=10&md5=fbec8bf576e2
f2cc65e0a364e632e3e8
Introduction:
Mismatch negativity (MMN) is an event-related potential measure of auditory
change detection. It is widely reported to be smaller in patients with
schizophrenia and may not improve along with otherwise successful clinical
treatment (Oades et al., 1997). The main aim of
this report is to explore ways of measuring and presenting four features
of frequency-deviant MMN dipole sources (dipole moment, peak latency,
brain location and orientation) and to relate these to the processes
of psychopathology and illness progression.
We already published a study of normal
healthy subjects showing thew automatic processing sources
for MMN bilaterally in the temporal lobes, left anterior cingulate and
right inferiror frontal cortex (MMN,
Jemel et al., 2002). [For sources of conscious controlled processing
illustrated by the Negative Difference (Nd), see Jemel
et al., 2003]
Methods:
Data from early onset patients (EOS) at the start of the illness in
adolescence, and others who had their first break in adolescence 15
years ago (S-15Y) were compared with two groups of age-matched healthy
controls (C-EOS, C-15Y). Putative generators in the 120-180 ms post-stimulus
latency ranges were modelled with brain electrical source analysis (BESA)
and mapped to the modified Montreal brain-atlas (Garneron algorhythm).
Results:
First: A 4-source model fitted the MMN
waveform recorded from all 4 groups, whether MMN amplitude was more
(EOS) or less (S-15Y) reduced.
Second: The locations
were in the left superior temporal and anterior cingulate gyri, right
superior temporal and inferior/mid frontal cortices.
Third: Dipole latencies
confirmed a bottom-up sequence of processing and dipole moments were
larger in the temporal lobes and on the left.
Fourth: Patients
showed small dipole location changes that were more marked in the S-15Y
than the EOS group (more rostral for the left anteriorcingulate, more
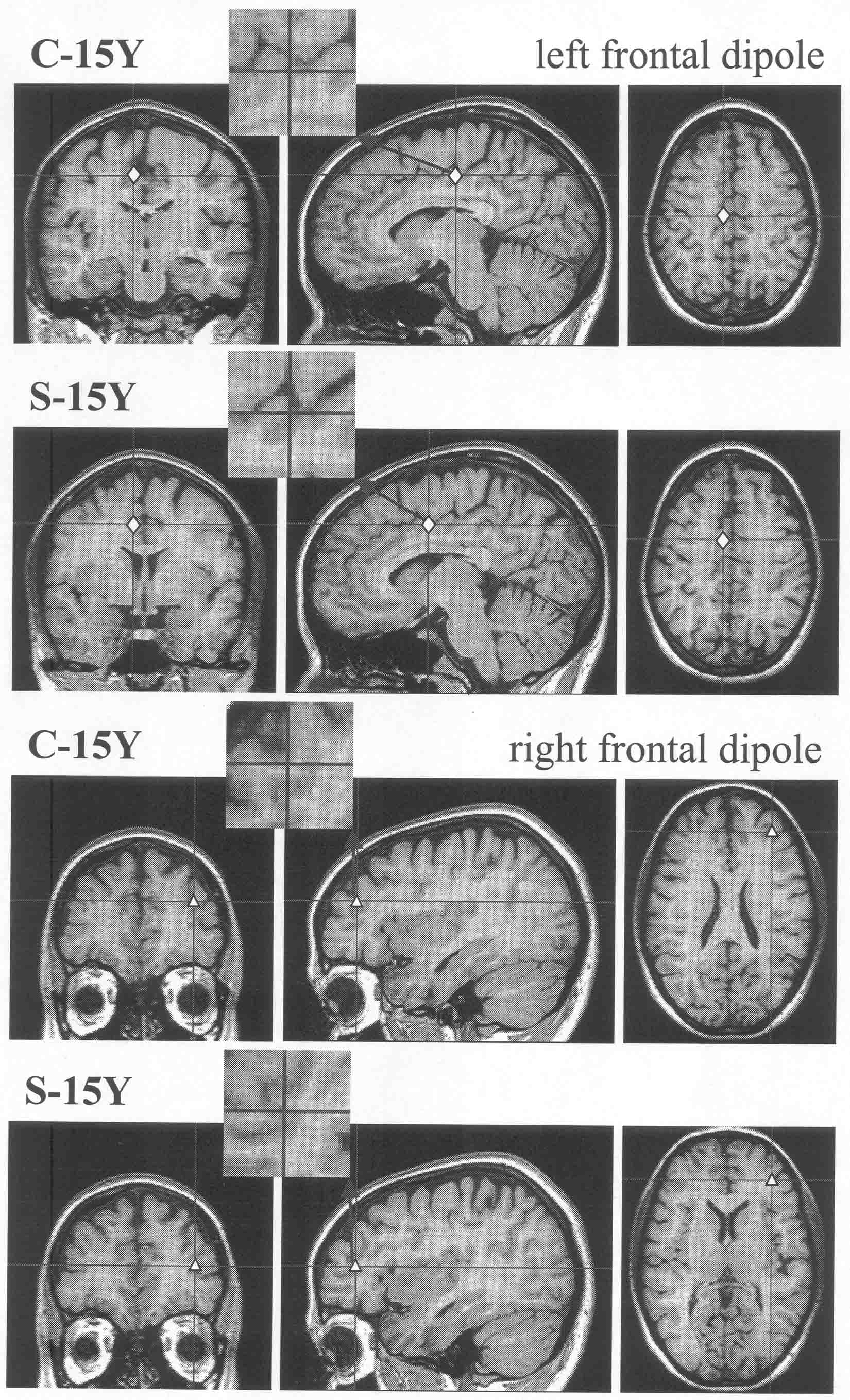
caudal for the right mid-frontal dipole) consistent with illness progression.
Conclusions:
The modelling of MMN dipole sources on brain atlas and anatomical images
suggests that there is a degree of dissociation during illness between
small progressive anatomical changes and some functional recovery indexed
by scalp recordings from patients with an onset in adolescence 15 years
before compared to adolescents in their first episode.
Fig. 1 below left: left
cingulate & right inferior/mid frontal sources (group solution)
on a control brain image
. . . . . . . . . . . . . . . . . . . . . . .
. . . . . . . . . . . . . . . . . . .. . . . . Fig.
2 below right: frontal and temporal lobe sources (mean of individual
solutions)
|

